eCOA and IRT Technology for the Realities of Rare Disease Research.
Rare disease clinical trials face challenges unlike any other study type. Small patient populations, geographic dispersion, heterogeneous symptom presentation, and limited natural history data require eCOA and IRT systems that are flexible, scalable, and able to adapt as scientific understanding evolves.
YPrime empowers global rare disease programs with eCOA, IRT, and eConsent solutions to improve data quality, reduce participant burden, and maintain inspection readiness. With support in 100+ countries and 250+ languages, YPrime empowers rare disease teams to execute globally without compromising data integrity.

Why Rare Disease Trials Require Specialized Clinical Trial Technology.
With patient populations often numbering in the hundreds (or less), studies must recruit across countries and regions, adapting to evolving endpoint knowledge, and managing variability in disease presentation and progression. Many conditions lack robust natural history data, requiring refinement of endpoints and assessment schedules as insights emerge.
Rare disease clinical trial technology must:
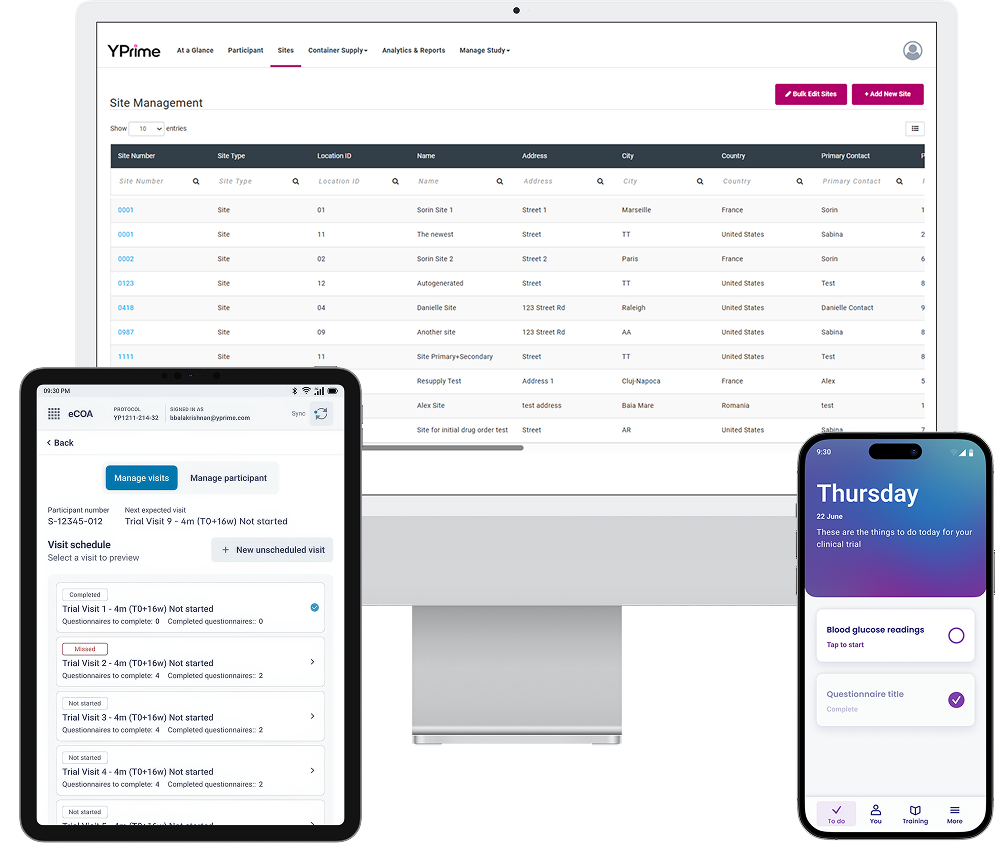
Technology Designed for the Rare Disease Participant Experience.
Rare disease trials place significant operational pressure on sites and study teams, particularly when managing dispersed populations and frequent protocol adjustments. YPrime eCOA and IRT support real-time oversight across both participant data collection and trial logistics, enabling:

Proven in Global Rare Disease Studies
YPrime supports rare disease clinical trials across a wide range of conditions with experience in ultra-small cohort studies and pediatric populations.
In one rare disease study, a top 10 pharmaceutical company approached YPrime for support on a Phase 3 trial, with fewer than 15 participants across less than five sites. With such a limited participant pool, every data point needed to be captured perfectly, with no room for missed entries.
YPrime deployed an eCOA solution tailored to the trial’s complex requirements, with an integrated blood glucose monitor, along with study drug, insulin, and symptom diaries. Notification pop-ups were embedded, prompting participants to complete symptom- triggered diaries, based on monitor readings.
The study used provisioned devices and remote data capture, crucial for reaching patients beyond sites. The study design emphasized patient engagement through alerts that boosted diary adherence, a critical factor in small cohort studies.
Explore Rare Disease Resources
FAQs
Rare disease trials involve small, dispersed populations, heterogeneous symptoms, and evolving endpoint definitions. Specialized eCOA and IRT technology helps sponsors adapt to these realities while maintaining data integrity, operational flexibility, and consistent data capture. It also supports global execution requirements, including localization, regulatory compliance, and caregiver reporting.
Challenges include limited natural history data, high geographic dispersion, frequent protocol changes, and heavy caregiver involvement. These factors require technology that supports flexibility, decentralization, and multiple reporting sources. Rare disease trials also require standardization across regions while accommodating local healthcare systems and regulatory requirements.
Many rare disease participants—especially pediatric patients—depend on caregivers. Proxy and observer reporting capabilities ensure continuity of data collection while reducing participant burden. Caregiver-friendly reminders and training materials also support accurate completion of more complex assessments.
By minimizing burden through adaptive scheduling, intuitive interfaces, and remote participation options, eCOA helps improve engagement and retention in trials where every participant is critical. Additionally, eCOA alerts can help address missing data quickly in small populations.



