The top clinical trial technology challenges sponsors face…
62% say timeline/delays
are the #1 issue
Many clinical trials fall behind schedule1
50% say data-change capabilities are the
#2 problem
Limited flexibility puts data quality at risk1
41% say protocol amendments are the #3 concern
Frequent amendments disrupt trial execution1
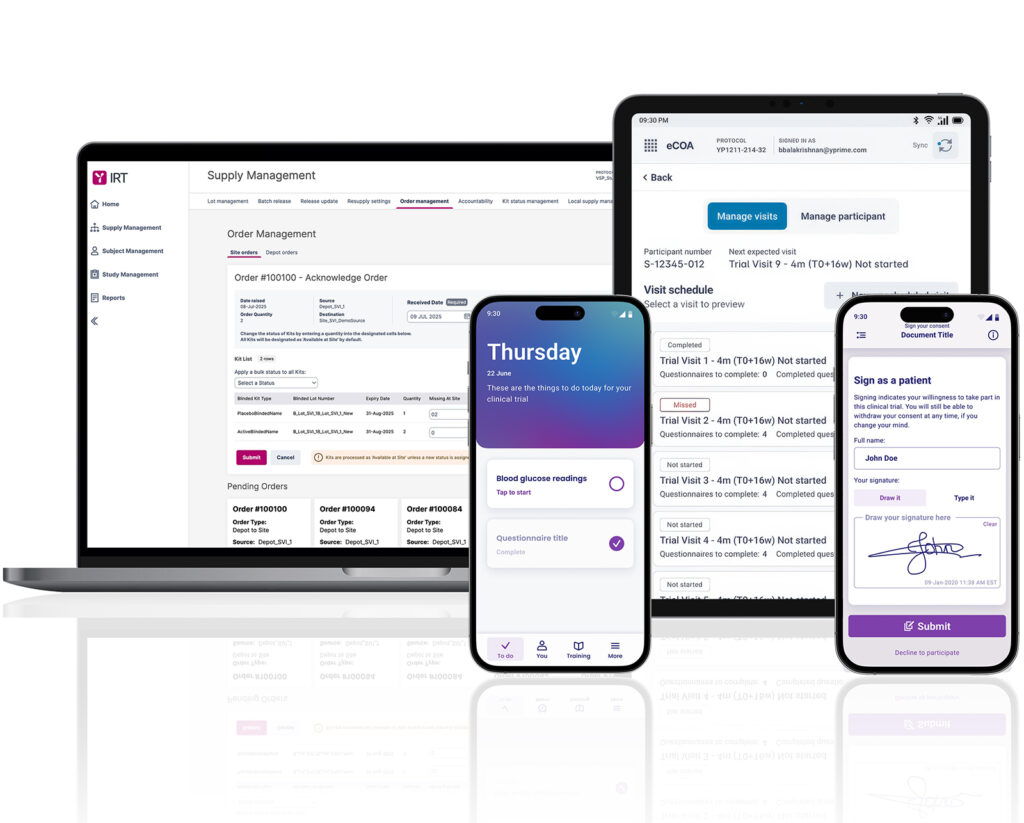
A Clinical Trial Technology Platform—Built for Speed, Flexibility, and Certainty
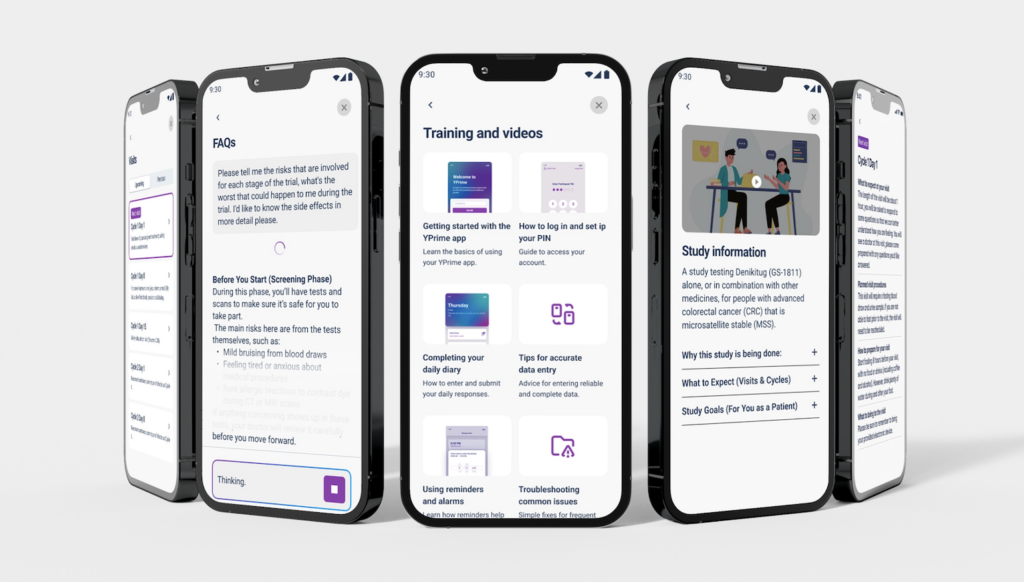
YPrime’s clinical trial technology platform offers patient engagement features across eConsent and eCOA, leveraging user-friendly design, personalization, and behavioral science to drive patient retention and adherence.
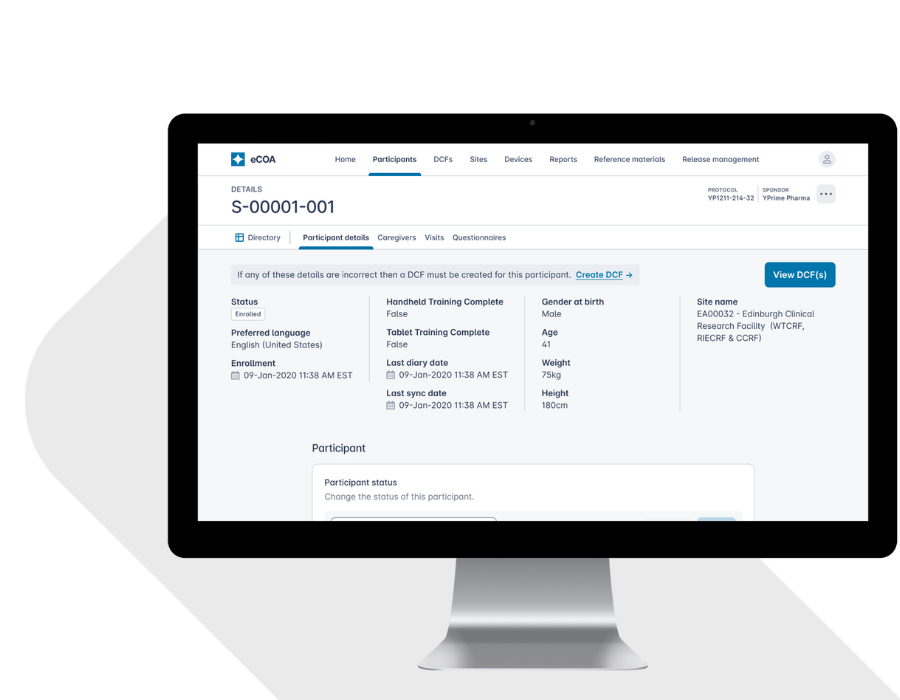
Clinical Trial Technology Experience That Matters.
~1,000
Studies Implemented
Globally
19+
Therapeutic
Areas
16+
Years eClinical Technology
and Services
We help pharma companies, emerging biotechs, and CROs simplify clinical trial technology, reduce site burden, and improve participant experience—without compromising data quality or compliance.
Explore Insights from Our Experts.
Gain valuable perspectives on clinical trial design, high-quality data capture, operational efficiencies, and, ultimately, how to solve with certainty in clinical research.
Source: