Upcoming Events and Webinars
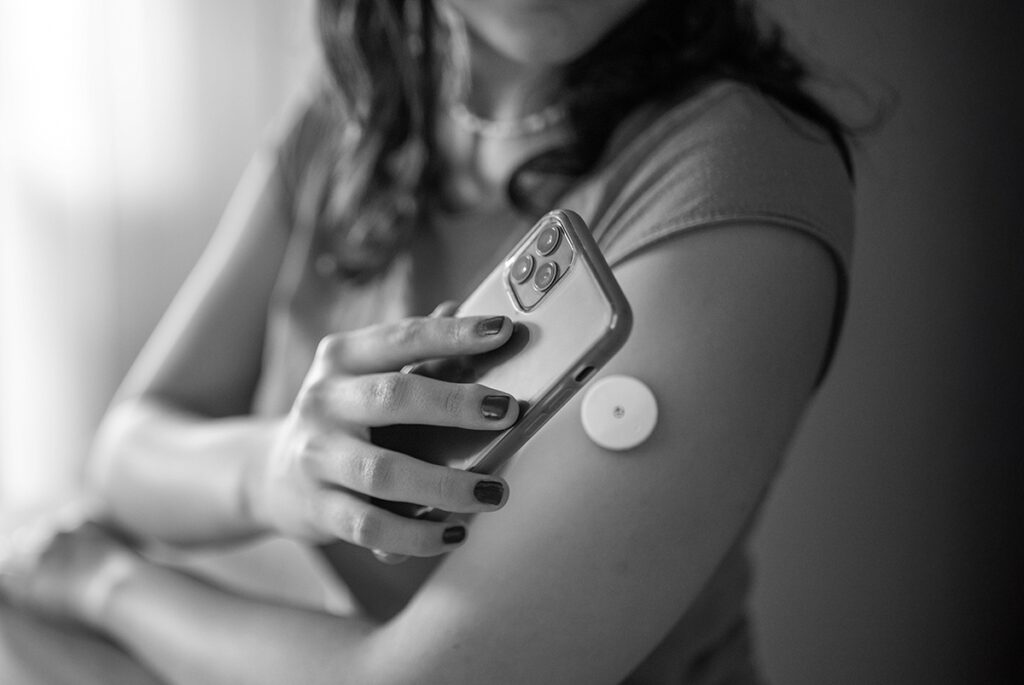
CGM or BGM? Evaluating the Right Glucose Monitoring Approach for Your Clinical Trial
Join YPrime experts for a focused discussion on choosing the right glucose monitoring strategy—BGM or…
CTS West Coast 2025
Will you be at CTS West Coast? Visit YPrime at booth #31 to explore how…
OCT Southern California 2025
Visit YPrime at OCT Southern California! Stop by booth #44 to discover how our solutions…
On-Demand Webinars
On-Demand Webinar: Let’s Talk eCOA: Three Innovations in 30 Minutes—And What It Means for You
YPrime’s Donna Mongiello and Aubrey Verna explore three eCOA innovations and their benefits for clinical…
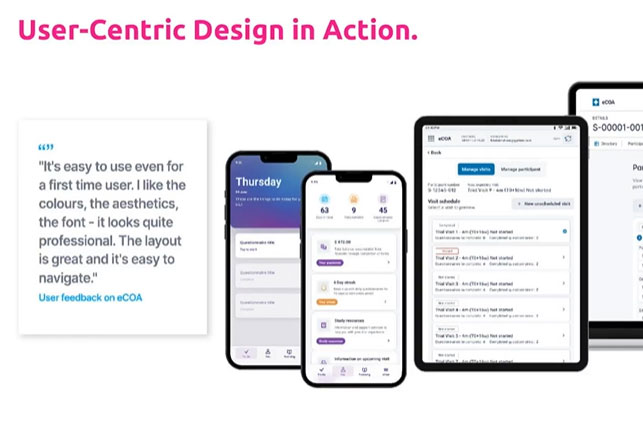
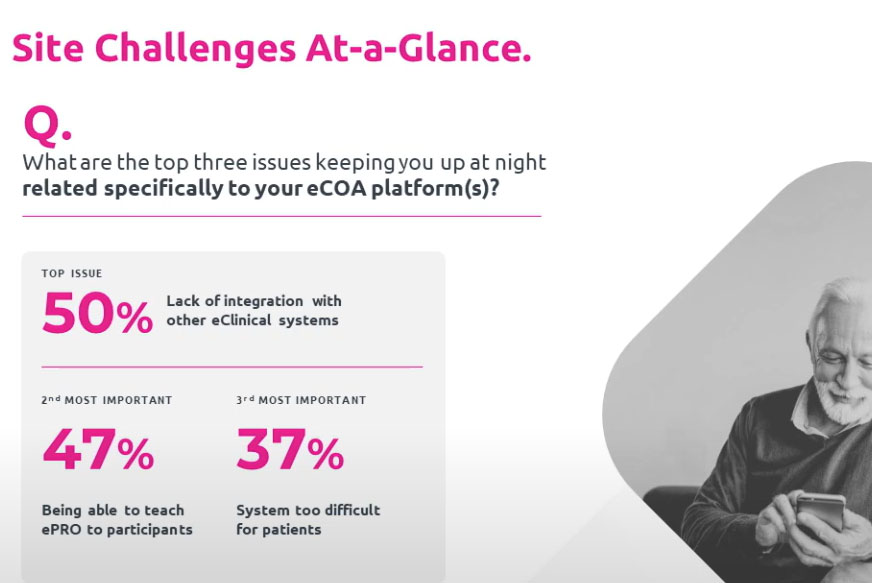
On-Demand Webinar: Boost Clinical Trials by Aligning Tech with User Needs
Industry leaders share insights for sponsors aiming to enhance clinical trial efficacy.
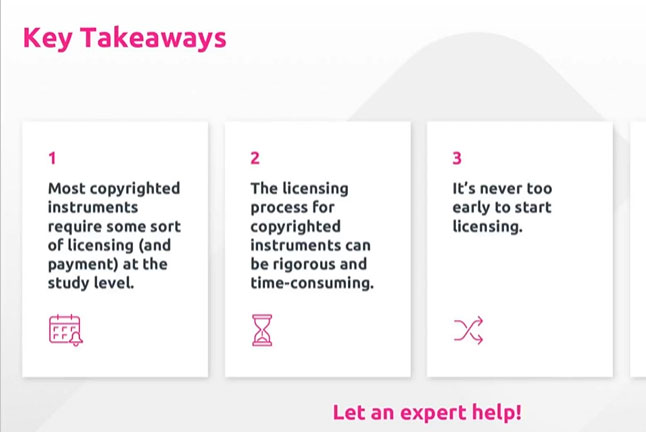
On-Demand Webinar: Building Your eCOA Foundation—Instrument Licensing and Translation
Get insights and strategies for navigating the complexities of eCOA instrument licensing and translation.